What is Wastewater?
Wastewater is sewage, stormwater, and water that have been used for various purposes around the community. Unless properly treated, wastewater can harm public health and the environment. Most communities generate wastewater from both residential and nonresidential sources
Residential wastewater
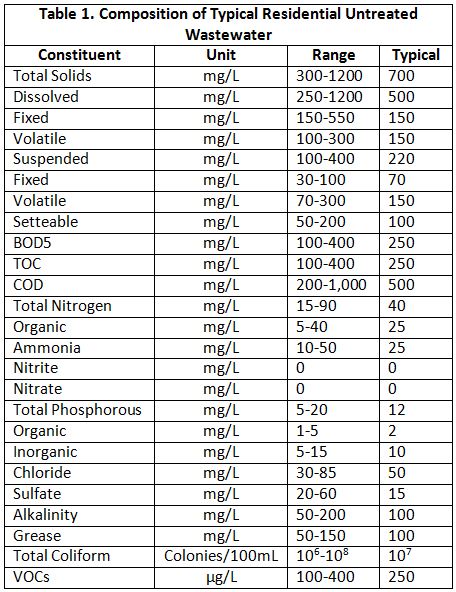
Although the word sewage usually brings toilets to mind, it actually is used to describe all types of wastewater generated from every room in a house. In the U.S., sewage varies regionally and from home to home based on such factors as the number and type of waterusing fixtures and appliances, the number of occupants, their ages, and even their habits, such as the types of foods they eat. However, when compared to the variety of wastewater flows generated by different nonresidential sources, household water shares many similar characteristics overall. “Table 1” lists the composition of typical residential untreated wastewater.
There are two types of domestic sewage: blackwater (wastewater from toilets) and graywater (wastewater from all sources except toilets). Blackwater and graywater have different characteristics, but both contain pollutants and disease-causing agents that require treatment.
Nonresidential wastewater
Nonresidential wastewater in small communities is generated by such diverse sources as offices, businesses, department stores, restaurants, schools, hospitals, farms, manufacturers, and other commercial, industrial, and institutional entities. Stormwater is a nonresidential source and carries trash and other pollutants from streets, as well as pesticides and fertilizers from yards and fields. Because of the variety of nonresidential wastewater characteristics, communities need to assess each source individually or compare similar types of nonresidential sources to ensure that adequate treatment is provided. For example, public restrooms may generate wastewater with some characteristics similar to sewage, but usually at higher volumes and at different peak hours. The volume and pattern of wastewater flows from rental properties, hotels, and recreation areas often vary seasonally as well.
Laundries differ from many other nonresidential sources because they produce high volumes of wastewater containing lint fibers. Restaurants typically generate a lot of oil and grease. It may be necessary to provide pretreatment of oil and grease from restaurants or to collect it prior to treatment, for example, by adding grease traps to septic tanks.
Wastewater from some nonresidential sources also may require additional treatment steps. For example, stormwater should be collected separately to prevent the flooding of treatment plants during wet weather. Trash and other large solids from storm sewers often are removed by screens.
In addition, many industries produce wastewater high in chemical and biological pollutants that can overburden treatment and community systems. Dairy farms and breweries are good examples — communities may require these types of nonresidential sources to provide their own treatment or preliminary treatment to protect community systems and public health.
What is in Wastewater?
Wastewater is mostly water by weight. Other materials make up only a small portion of wastewater, but can be present in large enough quantities to endanger public health and the environment. Because practically anything that can be flushed down a toilet, drain, or sewer can be found in wastewater, even household sewage contains many potential pollutants. The wastewater components that most wastewater facilities are designed to remove are suspended solids, biodegradable organics, and pathogenic organisms.
Inorganics
Inorganic minerals, metals, and compounds, such as sodium, potassium, calcium, magnesium, cadmium, copper, lead, nickel, and zinc are common in wastewater from both residential and nonresidential sources. They can originate from a variety of sources in the community including industrial and commercial sources, stormwater, and inflow and infiltration from cracked pipes and leaky manhole covers. Most inorganic substances are relatively stable and cannot be broken down easily by organisms in wastewater.
Large amounts of many inorganic substances can contaminate soil and water. Some are toxic to animals and humans and may accumulate in the environment. For this reason, extra treatment steps are often required to remove inorganic materials from industrial wastewater sources. Heavy metals, for example, which are discharged with many types of industrial wastewaters, are difficult to remove by conventional treatment methods. Although acute poisonings from heavy metals in drinking water are rare in the U.S., potential longterm health effects of ingesting small amounts of some inorganic substances over an
extended period of time are possible.
Inorganic ions in solution contribute to the conductivity (e.g., the ability to carry an electrical current) of the wastewater. The conductivity of domestic wastewater may be near that of the local water supply, although not necessarily. Backwash from water softeners contains high calcium chloride concentrations which will increase the conductivity.
Nutrients
Wastewater often contains large amounts of nitrogen and phosphorus in the form of nitrate and phosphate, respectively — nutrients that promote plant growth. Organisms only require small amounts of nutrients in biological treatment, so there normally is an excess of available nutrients in treated wastewater. In severe cases, excess nitrogen and phosphorous can result in eutrophication, the nutrient enrichment of water bodies causing excessive growth of aquatic plants (algae, cyanobacteria, rooted aquatic vegetation, duckweed). The dissolved oxygen in the water body becomes depleted when the aquatic plants die, fall to
the bottom, and are decomposed by aerobic bacteria. The oxygen depletion can reduce the populations of indigenous fish and other oxygen-consuming organisms.
Nutrients from wastewater have also been linked to ocean “red tides” that poison fish and cause illness in humans. Nitrogen in drinking water may contribute to miscarriages and is the cause of a serious illness in infants called methemoglobinemia or “blue baby syndrome.”
- Nitrogen –Nitrogen forms that are important in wastewater include organic nitrogen, nitrate (NO3–), nitrite (NO2–), ammonia (NH+4), and nitrogen gas (N2). All ofthese forms are biochemically interconvertible.
- Organic nitrogen is nitrogen bound to carbon. It is the principle nitrogen constituent in feces. Organic nitrogen also includes urea (H2NCONH2) which is the principal compound in urine. Organic nitrogen is not readily available to plants; it needs bacterial conversion to nitrate before it is available for plants.
- Nitrate is the most oxidized species of nitrogen. Nitrate is readily available to plants and is considered the limiting nutrient (the nutrient that keeps thebiotic system in balance) for primary productivity in salt waters, an important consideration in the design of treatment systems along coastal areas.Because nitrate, in solution, is a negative ion, it will not bind to soil which is also negatively charged. Therefore, nitrate passes through soil to groundwater which is why regulatory agencies may be requiring some treatment systems to provide nitrogen reduction in the effluent. Nitrate is also the species of nitrogen for which a limit has been set for drinking water (10mg/L) due to blue baby syndrome.
- Nitrite is not usually observed in water sources because it is readily converted to nitrate by bacterial processes; however, it is extremely toxic to most fish and other aquatic species. Also, nitrites are oxidized by chlorine and can, therefore, increase the chlorine dosage requirements and the cost of disinfection.
- Ammonia exists in water as either the ammonium ion (NH4+) or ammoniagas (NH3), depending on the pH of the water. At pH levels above 9.3, ammonia gas is the predominate form, and at pH levels below 9.3, the ammonium ion is the predominant form. Ammonia is usually present in surface water and is due to the chemical oxidation of urea and anaerobic processes. Ammonia, a positive ion, binds to soil which is negatively charged; therefore, ammonia is not readily leached from the soil. Plants can readily use the ammonia form of nitrogen.
- Phosphorus – Phosphorus also exists in wastewater in many forms and includes soluble orthophosphate ion (PO4-3), organically-bound phosphate, and other phosphorus/oxygen forms. Most of the organically-bound phosphate in wastewater is from excretia and food residue. Some cleaning agents still contain phosphate, although the practice of adding phosphate to cleaning agents has been vastly reduced since the 1980s. Phosphorus is usually the limiting nutrient in freshwater surface waters and is the principal cause of eutrophication in surface water bodies. However, phosphate rapidly combines with other naturally-occurring chemicals, such as limestone, to form calcium phosphate. If a subsurface effluent distribution system is close to a sensitive water body, limestone added to the soil absorption system can stop the phosphate from migrating to the water body.
Oil and grease
Oil and grease is the term given to the combination of fats, oils, waxes, and other related constituents found in wastewater. Fatty organic materials from animals, vegetables, and petroleum are not quickly broken down by bacteria and can cause pollution in receiving environments. When large amounts of oils and greases are discharged to receiving waters from community systems, they increase BOD levels, and they may float to the surface and harden, causing aesthetically unpleasing conditions. They also can trap trash, plants, and other materials causing foul odors and attracting flies and mosquitoes and other disease
vectors. In some cases, too much oil and grease causes septic conditions in ponds and lakes by preventing oxygen from the atmosphere from reaching the water.
If the wastewater is warm and greasy, the grease cannot quickly separate from the water, so the grease flows through the septic tank and into the soil where it solidifies upon cooling. The grease can clog the gravel and soil pores ruining the drainage system. Excessive grease also adds to the septic tank scum layer, requiring more frequent tank pumping. Both possibilities can result in significant costs to homeowners and are the reasons why grease traps are mandatory for restaurants and food service facilities.
Petroleum-based waste oils used for motors and industry are considered hazardous waste and should be collected and disposed of separately from wastewater.
Organic Matter
Organic materials are found everywhere in the environment. They are composed of the carbon-based chemicals that are the building blocks of most living things. Organic materials in wastewater originate from plants, animals, or synthetic organic compounds, and enter wastewater in human wastes, paper products, detergents, cosmetics, foods, and from agricultural, commercial, and industrial sources. Organic compounds normally are some combination of carbon, hydrogen, oxygen, nitrogen, and other elements. Many organics are proteins, carbohydrates, or fats and are biodegradable, which means they can be consumed and broken down by organisms. However, even biodegradable materials can cause pollution. In fact, too much organic matter in wastewater can be devastating to receiving waters.
Large amounts of biodegradable materials are dangerous to lakes, streams, and oceans because organisms use dissolved oxygen in the water to break down the wastes. This can reduce or deplete the supply of oxygen in the water needed by aquatic life, resulting in fish kills, odors, and overall degradation of water quality. The amount of oxygen organisms need to break down wastes in wastewater is referred to as the biochemical oxygen demand (BOD) and is one of the measurements used to assess overall wastewater strength.
Some organic compounds are more stable than others and cannot be quickly broken down by organisms, posing an additional challenge for treatment. This is true of many synthetic organic compounds developed for agriculture and industry. In addition, certain synthetic organics are highly toxic. Insecticides and herbicides are toxic to humans, fish, and aquatic plants and often are disposed of improperly in drains or carried in stormwater. In receiving waters, they kill or contaminate fish, making them unfit to eat. They also can damage processes in treatment plants. Benzene and toluene are two toxic organic compounds found
in some solvents, pesticides, and other products. New synthetic organic compounds are being developed all the time, which can complicate treatment efforts.
Pathogens
Many disease-causing viruses, parasites, and bacteria also are present in wastewater and enter from almost anywhere in the community. These pathogens often originate from people and animals that are infected with or are carriers of a disease. For example, graywater and blackwater from typical homes contain enough pathogens to pose a risk to public health. Other likely sources in communities include hospitals, schools, farms, and food processing plants.
Some illnesses from wastewater-related sources are relatively common. Gastroenteritis can result from a variety of pathogens in wastewater, and cases of illnesses caused by the parasitic protozoa Giardia lambia and Cryptosporidium are not unusual in the U.S. Other important wastewater-related diseases include hepatitis A, typhoid, polio, cholera, and dysentery. Outbreaks of these diseases can occur as a result of drinking water from wells polluted by wastewater, eating contaminated fish, or recreational activities in polluted waters. Some illnesses can be spread by animals and insects that come in contact with wastewater.
Even municipal drinking water sources are not completely immune to health risks from wastewater pathogens. Drinking water treatment efforts can become over-whelmed when water resources are heavily polluted by wastewater. For this reason, wastewater treatment is as important to public health as drinking water treatment.
Solids
Solid materials in wastewater can consist of organic and/or inorganic materials and organisms. The solids must be significantly reduced by treatment or they can increase BOD levels when discharged to receiving waters and provide places for microorganisms to escape disinfection. They also can clog soil absorption fields in treatment systems. The amount of solids in the wastewater affects the septic tank size and the frequency for pumping out the septic tank. Proper solids analysis is important for the control of biological and physical wastewater treatment processes and assess compliance with effluent quality limits.
- Total solids – The term applied to the residue left after evaporation; it includes the total suspended solids and total dissolved solids.
- Suspended solids – The portion of wastewater that, when passed through a filter, remains on the filter. Materials that resist settling may remain suspended in wastewater. Suspended solids in wastewater must be treated, or they will clog soil absorption systems or reduce the effectiveness of disinfection systems.
- Dissolved solids – The portion of wastewater that, when passed through a filter, does not remain on the filter. Small particles of certain wastewater materials can dissolve like salt in water. Some dissolved materials are consumed by microorganisms in wastewater, but others, such as heavy metals, are difficult to remove by conventional treatment. Excessive amounts of dissolved solids in wastewater can have adverse effects on the environment.
- Settable solids – Certain substances, such as sand, grit, and heavier organic and inorganic materials, settle out from the rest of the wastewater stream during the preliminary stages of treatment. On the bottom of settling tanks and ponds, organic material makes up a biologically active layer of sludge that aids in treatment. Knowing the amount of setteable solids in the wastewater provides information on how much sludge will be created in the septic tank.
- Fixed solids – The term applied for the residue (total, suspended, or dissolved) that is left after igniting a dried sample. The weight loss on ignition is called volatile solids. Each type of solids is processed differently, so it is essential to understand the distinction amongst the various types. Setteable and some suspended solids are usually removed by filtration or settling. Other suspended solids and dissolved solids are treated by biological processes or chemical precipitation.
Other Important Wastewater Characteristics
In addition to the many substances found in wastewater, there are other characteristics that system designers and operators use to evaluate wastewater. For example, color, odor, and turbidity of wastewater give clues about the amount and type of pollutants present and treatment necessary. The following are some other important wastewater characteristics that can affect public health and the environment, as well as the design, cost, and effectiveness of treatment.
Temperature
The temperature of wastewater is usually higher than that of the water supply because of the addition of warm water from domestic use. Wastewater temperature is important for two reasons: (1) biological processes are temperature dependent and (2) chemical reactions and reaction rates and aquatic life are all temperature sensitive.
The best temperatures for wastewater treatment range from 77 to 95 °F. In general, biological treatment activity accelerates in warm temperatures and slows in cool temperatures but extreme hot or cold can stop treatment processes altogether. For example, aerobic digestion and nitrification stop when temperatures rise to 122 °F; and when temperatures fall below 36 °F, virtually all biological activity becomes dormant. Therefore, some systems are less effective during cold weather and some may not be appropriate for very cold climates.
Wastewater temperature also affects receiving waters. Hot water, for example, which is a byproduct of many manufacturing processes, can be a pollutant. When discharged in large quantities, it can raise the temperature of receiving streams locally and disrupt the natural balance of aquatic life.
pH
The acidity or alkalinity of wastewater affects both treatment and the environment. The pH is actually the measure of the inverse concentration of hydrogen ions and is a logarithmic scale. As pH values lower, it indicates increasing acidity; while a high pH indicates alkalinity (a pH of 7 is neutral). The pH of wastewater needs to remain between 6 and 9 to protect beneficial organisms. Acids, cleaning agents, and other substances that alter pH can inactivate treatment processes when they enter wastewater.
Gases
Certain gases in wastewater can cause odors, affect treatment, or are potentially dangerous. Methane gas, for example, is a byproduct of anaerobic biological treatment (found in septic tanks) and is highly combustible. Special precautions need to be taken near septic tanks, manholes, treatment plants, and other areas where wastewater gases can collect. Hydrogen sulfide and ammonia gases can be toxic and pose asphyxiation hazards. Ammonia as a dissolved gas in wastewater also is dangerous to fish. Both gases emit odors, which can be a serious nuisance. Unless effectively contained or minimized by design and location, wastewater odors can affect the mental well-being and quality of life of residents. In some cases, odors can even lower property values and affect the local economy.
Flow
Whether a system serves a single home or an entire community, it must be able to handle fluctuations in the quantity and quality of wastewater it receives to ensure proper treatment is provided at all times. Systems that are inadequately designed or hydraulically overloaded may fail to provide treatment and allow the release of pollutants to the environment. To design systems that are both as safe and as cost-effective as possible, engineers must estimate the average and maximum (peak) amount of flows generated by various sources.
Because extreme fluctuations in flow can occur during different times of the day and on different days of the week, estimates are based on observations of the minimum and maximum amounts of water used on an hourly, daily, weekly, and seasonal basis. The possibility of instantaneous peak flow events that result from several or all water-using appliances or fixtures being used at once also is taken into account.
The number, type, and efficiency of all water-using fixtures and appliances at the source are factored into the estimate (for example, the number and amount of water normally used by faucets, toilets, and washing machines), as is the number of possible users or units that can affect the amount of water used (for example, the number of residents, bedrooms, customers, students, patients, seats, or meals served).
According to studies, water use in many homes is lowest from about midnight to 5 a.m., averaging less than one gallon per person per hour, but then rises sharply in the morning around 6 a.m. to a little over 3 gallons per person per hour. During the day, water use drops off moderately and rises again in the early evening hours.
Weekly peak flows may occur in some homes on weekends, especially when all adults work during the week. In U.S. homes, average water use is approximately 45 gallons per person per day, but may range from 35 to 60 gallons or more.
Peak flows at stores and other businesses typically occur during business hours and during meal times at restaurants. Rental properties, resorts, and commercial establishments in tourist areas may have extreme flow variations seasonally.
Estimating flow volumes for centralized treatment systems is a complicated task, especially when designing a new treatment plant in a community where one has never existed previously. Engineers must allow for additional flows during wet weather due to inflow and infiltration of extra water into sewers. Excess water can enter sewers through leaky manhole covers and cracked pipes and pipe joints, diluting wastewater, which affects its overall characteristics, and increasing flows to treatment plants sometimes by as much as three or four times the original design load.
Testing and Measuring Wastewater
System operators, designers, and regulatory agencies use tests to evaluate the strength of wastewater and the amount of treatment required, the quality of effluent at different stages of treatment, and the quality of receiving waters at the point of discharge. Tests also determine whether treatment is in compliance with state, local, and federal regulations. In small communities, operators and health officials often are trained to collect samples and perform some or all wastewater tests themselves. An option that sometimes is more economical for small systems is to send samples away to a lab for testing. The following area few important tests:
BOD – biochemical oxygen demand
The BOD test measures the amount of dissolved oxygen organisms are likely to need to degrade wastes in wastewater. This test is important for evaluating both how much treatment wastewater is likely to require and the potential impact that it can have on receiving waters.
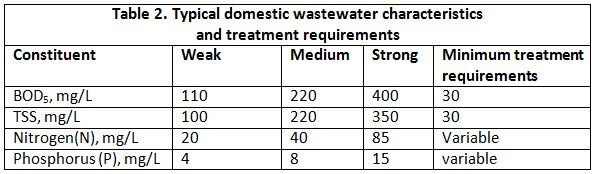
To perform the test, wastewater samples are placed in BOD bottles and are diluted with specially prepared water containing dissolved oxygen. The dilution water is also “seeded” with bacteria when treated wastewater is being tested. The amount of dissolved oxygen in the diluted samples is measured using a dissolved oxygen meter, and the samples are then stored at a constant temperature of 20 degrees Celsius (68 degrees Fahrenheit). Common incubation periods are five, seven, or twenty days; five days (or BOD5) is the most common. At the end of the incubation period, the dissolved oxygen is measured again. The amount that was used (expressed in milligrams per liter) is an indication of wastewater strength. Refer to the Table 2 for some typical BOD amounts. For an in-depth explanation of BOD and testing wastewater, refer to the module “Fundamental Microbiology of Sewage”.
TSS – total suspended solids
In addition to BOD, estimating the amount of suspended solids in wastewater helps to complete an overall picture of how much secondary treatment is likely to be required. It also indicates wastewater clarity and is important for assessing the potential impact of wastewater on the environment.
After large solids are removed in primary treatment, TSS is measured as the portion of solids retained by a 2.0-micron filter. Refer to the Table 2 for some typical TSS amounts.
TC and FC – total coliforms and fecal coliforms
Coliform tests are useful for determining whether wastewater has been adequately treated and whether water quality is suitable for drinking and recreation. Because they are very abundant in human wastes, coliform bacteria are much easier to locate and identify in wastewater than viruses and other pathogens that cause severe diseases. For this reason, coliform bacteria are used as indicator organisms for the presence of other, more serious pathogens. Some coliforms are found in soil, so tests for fecal coliforms are considered to be the most reliable. However, tests for both total coliforms and fecal coliforms are commonly used. There are two methods for determining the presence and density of coliform bacteria. The membrane filter (MF) technique provides a direct count of colonies trapped and then cultured. The multiple tube fermentation method provides an estimate of the most probable number (MPN) per 100 milliliters from the number of test tubes in which gas bubbles form after incubation.
 .
.
Please note that this information was adapted from the Northern Arizona University
Department of Civil and Environmental Engineering.







